Icad fda approval

In fact, the FDA has been reviewing technology with AI components (e.g., rule-based systems, machine learning) for more than a decade. However…It would be useful for the agency to offer meaningful reference to machine learning or deep learning among the examples of potential use cases. Nearly all AI will remain under FDA oversight. The suggestion that the FDA did not address the topic becasue it failed to explicitly mention AI within the document shows the challenges for those unfamiliar with understanding this complex subject. The FDA does use the word “algorithm” in its guidance, and although algorithms can vary in sophistication, much of today’s AI technology is based on algorithmic intelligence. However, it is unnecessary for the agency to use the term “AI” to provide guidance on how it will consider associated technologies and use cases. Indeed, a “Command+F” search for “Artificial Intelligence” returns zero results. Following the release, STAT News mentioned experts were disappointed because the agency gave no insight into how it views artificial intelligence. Last week, the FDA finally released its long-awaited Draft Guidance on Clinical Decision Support. Medical applications that rely on “black box” algorithms unable to be fully understood by the end-user (basically all AI) will be regulated, posing challenges for AI adoption.
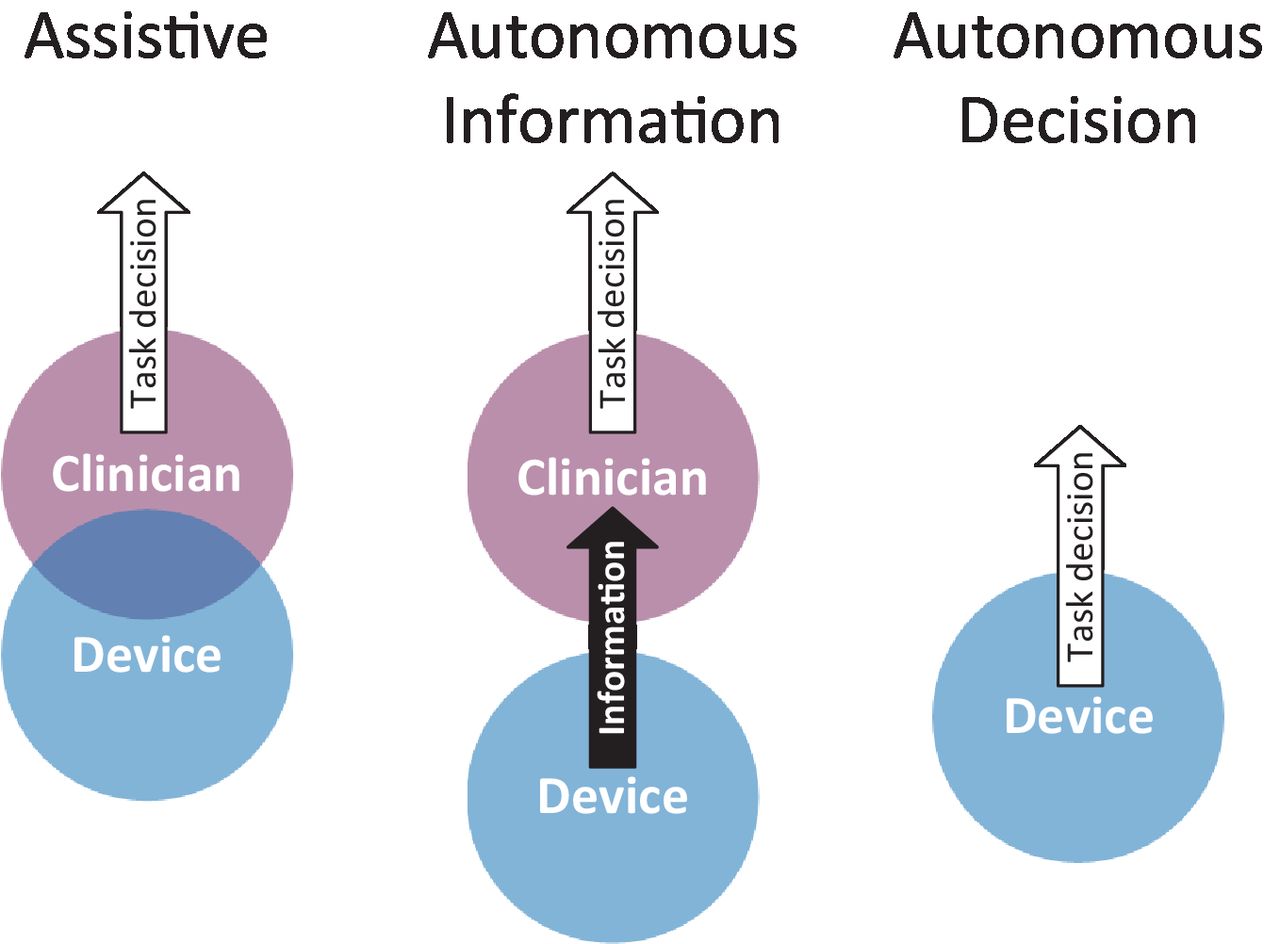
For applications with data originating from medical devices, the FDA will continue its oversight, AI or not (e.g., medical image processing).The FDA has released long-anticipated draft guidance on how they intend to regulate clinical decision support products.
